Medical device industry
Medical device manufacturing with proactive quality management in compliance with ISO 13485
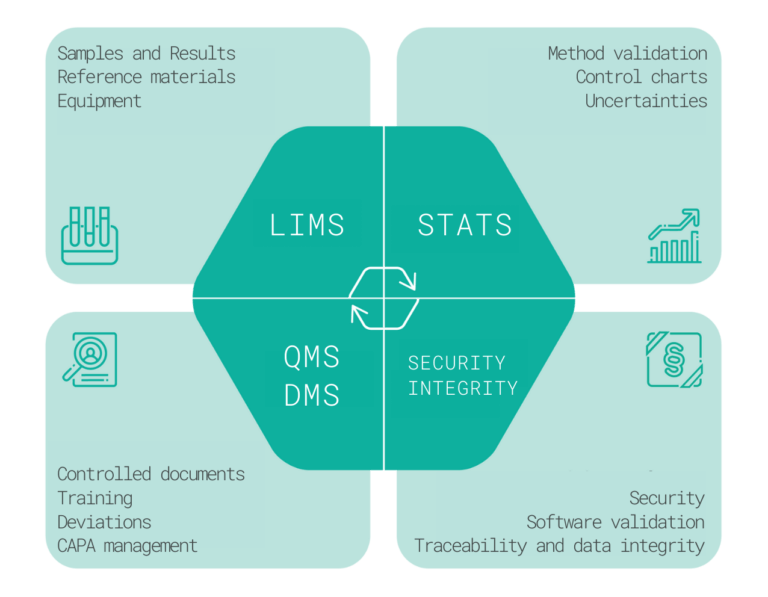
Characteristics and functions
EffiChem was developed in compliance with the harmonized GMP requirements for US, EU and JP, Vol. 4 of EU GMP, Supplement 11 (Annex 11), 21 CFR Part 11, according ISO 13485 and GAMP 5 for validation of computerized systems.
Document management and training is an intrinsic part of the Quality Management System for medical device industries. It includes the complete process from document creation, review, and approval, to activation/release, periodic revisions, archiving, or document cancellation.
Training management is linked to the documentation, followed with the creation of Tests, test questions, and the correct answers, the ability to record and evaluate the user answers, calculate the test scores, initiate re-testing, monitor due dates, and remind and escalate.
For Document Management and Training, workflows can be configured according to customer requirements and the desired process. Workflows can be enhanced with email notifications, action management, escalation, and other general functionalities, including managerial reporting.
The QMS modules can be used for record keeping and quality management. It can include risk management, vendor quality management, change control, audits and observation handling, evaluation and handling of nonconformances, CAPA management, complaint handling and reporting, and other specific modules required by the customer.
Workflows are configured according to your user requirements and the process. Workflows can be enhanced with action management, email notifications incl. escalations, and other general functionalities, including managerial reporting.
In case the medical device manufacturer runs a quality control laboratory, LIMS can be used to manage the lab processes and to ensure the related quality records. The main process is the sample testing, recording results and comparing them with the specifications. Besides, LIMS can cover the equipment management, incl. calibrations and maintenance, reference materials management, and other laboratory agendas and record keeping such as environmental monitoring and data logging.
LISM supports bar codes and QR codes and can be interfaced with equipment and ERP systems (under preparation).
Statistical Data Evaluation typically includes the validation of test methods, estimation of uncertainties, development and use of control charts, trending, creation and use of calibration models, and evaluation of inter-laboratory studies.
When there are multiple options for statistical evaluation of data, all options are included in the software, e.g. for a correct evaluation of Accuracy, options include a t-test and a statistical regression evaluation; for linearity, there is the correlation coefficient calculation, ANOVA or Sign test. The output of statistical data evaluation is a numerical result, a graph, and an automatically generated report.
Customer project support
EffiValidation 4.0



